Each orbital has a capacity of two. The electronic structure of carbon.
Orbital diagram of boron (b) 6:.

Orbital diagram of carbon. In the above orbital diagram, the box represents an orbital. Orbital diagram of beryllium (be) 5: Caulton and fenske began with a molecular orbital diagram for co (see fig.
Dot diagrams are very different to orbital diagrams, but they’re still very easy to understand. Located in the ii period. The maximum number of electrons allowed in an orbital is 2, each with opposite spins (pauli's.
Electron configuration of carbon in the excited state atoms can jump from one orbital to another in an excited state. How do you write the orbital diagram for carbon? In this diagram (and the orbital diagrams that follow), the nucleus is shown very much larger than it really is.
Carbon is the sixth element with a total of 6 electrons. The orbital diagram for carbon is drawn with 3 orbitals. Electronic configuration of the carbon atom:
The below mention diagram is the valence shell electronic configuration of both the carbon and. The orbital diagram shows how the electrons are arranged within each sublevel. 1s 2 2s 2 2p 2.
Orbital diagram of hydrogen (h) 2: The hybridization of carbon monoxide is sp as its geometrical structure is linear. The orbitals are 1s, 2s, and 2p.
Orbital diagram of helium (he) 3: They consist of the symbol for the element in the center, surrounded by dots. This is just for clarity.
The carbon orbital diagram contains 2 electrons in the 1s orbital, 2 electrons in the 2s orbital, and. A molecular orbital diagram, or mo diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination. In writing the electron configuration for carbon the first two electrons will.
Carbon has 2 electrons in its first shell and 4 in its second shell.check me out: Orbital diagram is the filling of the electrons into different orbitals according to the number of electrons present in an atom and an orbital consists of a maximum of two electrons. [he] 2s 2 2p 2.
Orbital diagram of lithium (li) 4: Molecular orbital diagram of carbon molecule video lecture from chapter nature of chemical bond of subject chemistry class 11 for hsc, iit jee, cbse & neet.w. This is clearly shown in the figure of the orbital diagram of carbon.
Hence, draw the blank orbital diagram of carbon upto 2p subshell as follows:
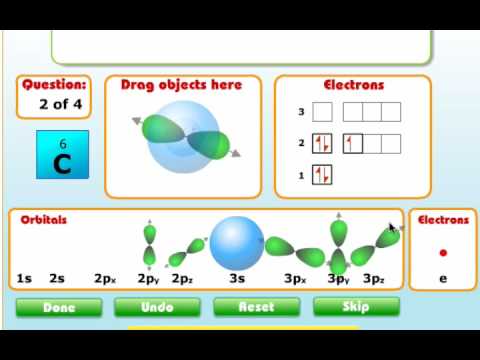
Orbital Diagram Carbon Quantum Theory Hund's Rule Aufbau Principle And Pauli Exclusion Principle - Youtube
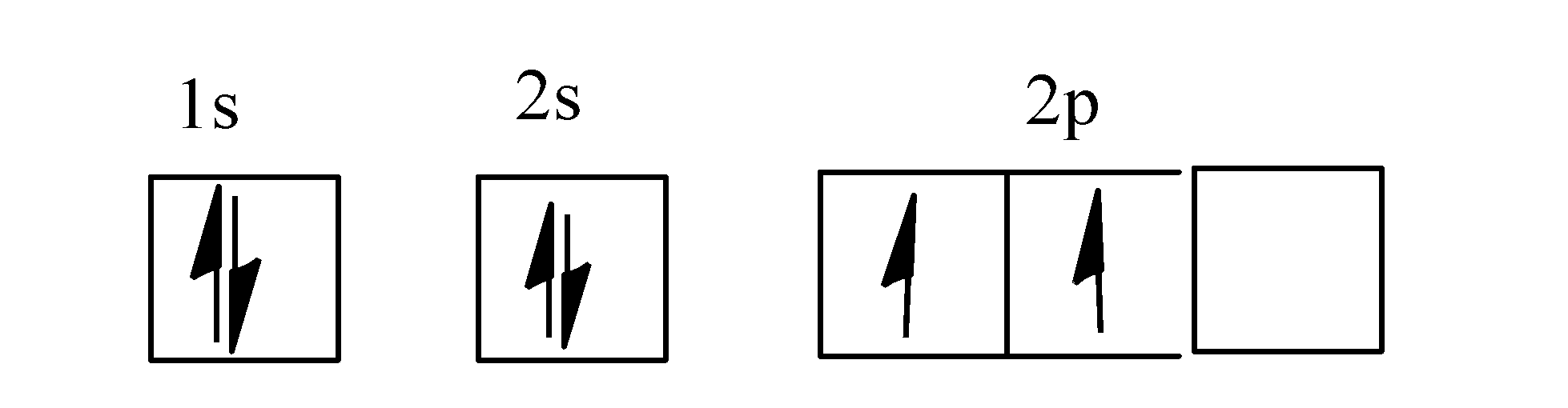
How Do You Write The Orbital Diagram For Carbon Class 11 Chemistry Cbse
