If you were writing an electron configuration for silicon using the noble gas notation, which noble gas would you use? From the expression it is visible that the 4s orbit of zinc consists of 2 electrons.

List Of Electron Configurations Of Elements
The study literally configurations for a given elements.

Noble gas configuration for silicon. Investigating first interstitial configurations, the tetrahedral t site is the most stable site for helium with e i = 1.00 ev (table 1 and fig. The ground state electron configuration of silicon is 1s 2 2s 2 2p 6 3s 2 3p 2. This problem has been solved!
Orbital diagram, electron configuration, and the noble gas notation for a silicon (si) atom. The orbitals are p x, p y, and p z and each orbital can have a maximum of two electrons. Using noble gas notation write the electron configuration for the silicon atom.
So for sodium, we make the substitution of [ne] for the 1 s2 2 s2 2 p6 part of the configuration. 1s 2 2s 2 2p 6 3s 2 3p 2 to write the noble gas configuration, find the noble gas that is just before silicon on the periodic table. Since 1s can only hold two electrons the next 2 electrons for silicon go in the 2s orbital.
They are helium, neon, argon, krypton, xenon, and radon. So for sodium, we make the substitution of [ ne] for the 1 s 2 2 s 2 2 p 6 part of the configuration. The nearest noble gas to this element is neon.
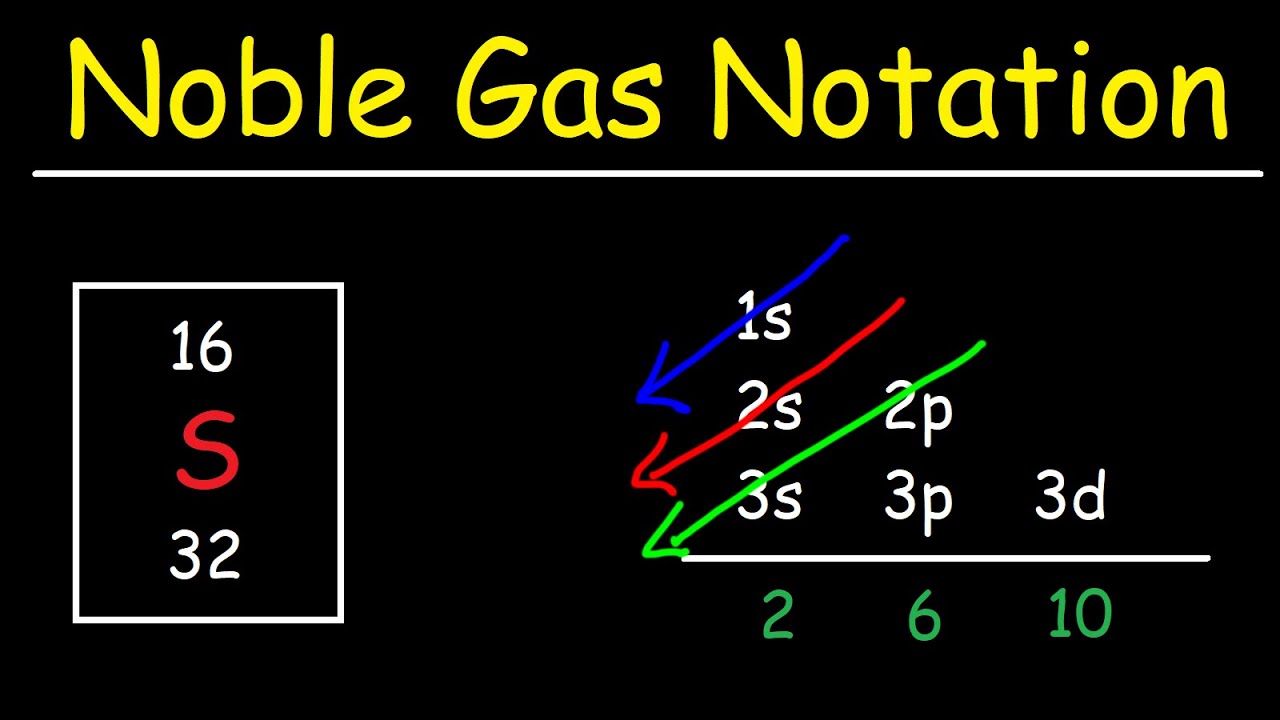
And we know that iss b d f thesis orbital's can hold maximum off to six, 10 and 14 electrons. A noble gas configuration of an atom consists of the elemental symbol of the last noble gas prior to that atom, followed by the configuration of the remaining electrons. The nex six electrons will go in the 2p orbital.
The p orbital can hold up to six electrons. For silicon, all but the outermost shell is filled. Silicon is right below carbon in the periodic table, and we can write its full electron configuration, just for comparison, so if we start up here, with hydrogen, silicon has the electron configuration 1s2 2s2 2p6, so as a reminder, these are our s electrons because they're in s orbitals.
In writing the electron configuration for silicon the first two electrons will go in the 1s orbital. So the number of filled shells is 2. Noble gas notation is defined as the notation in which the configuration of an atom is written in terms of previous noble gas.
The complete electron configuration for the chromium (iii) ion is 1s22s22p63s23p64s03d3. Using the diagonal rule, the electron configuration for silicon looks like this: The noble gas notation for silicon atom is explanation:
So the round district electron configuration, often atom, describes the lowest energy or evidence that its electrons are hoping. Hey, guys needs to problem 53. A noble gas configuration of an atom consists of the elemental symbol of the last noble gas prior to that atom, followed by the configuration of the remaining electrons.
[ne] 3s23p2 the full electron configuration of silicon is 1s22s22p63s23p2. Silicon is the 14th element of the periodic table having 14 electrons. The outer configuration is 5s^2 4d^2.
Its electronic configuration is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10. Silicon (si) has 14 electrons. To find its noble gas shorthand, look at the full electron configuration and see how many filled shells there are.
See the answer see the answer see the answer done loading It can be expressed as [ar] 3d 10 4s 2 with reference to the configuration of the nearest noble gas. 2 ,8 ,4 is the answer what is the noble gas configuration for silicon 2
Sodium’s noble gas configuration becomes [ne]3 s1. We need to write down the ground.
Electron Configuration With Noble Gas Notation - Youtube

Electron Configuration Topic #8 September How Was The Atomic Model Developed? - Ppt Download