A faster way to determine how many pi bonds the molecule has is to know that a double bond is comprised of 1 sigma. Co2 sigma and pi bonds there are two double bonds in carbon dioxide.

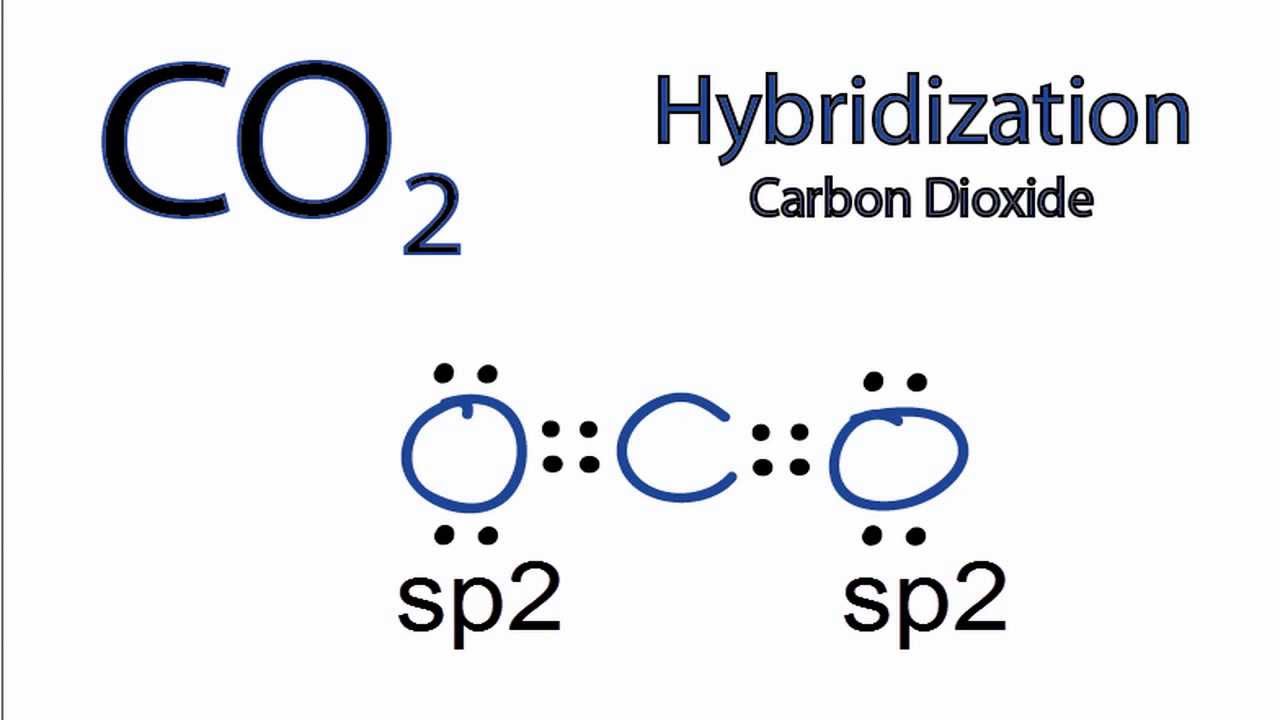
Hybridisation And Bonding In Carbon Dioxide - Youtube
Pi or π bonds occur when there is an overlap between unhybridized p orbitals of two adjacent atoms.
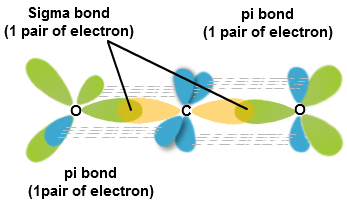
Co2 pi and sigma bonds. Explain how the sigma and pi bonds are made in co2. How many sigma and pi bonds does co2 have. Sigma and pi bonds are an aspect of valence bond theory and explains the existence of double and triple bonds.
Therefore, co2 has 2 pi bonds and 2 sigma bonds. Here in co2, both oxygen atoms form. From the molecular orbital diagram of c2 we get that the bond order in 2.
A faster way to determine how many pi bonds the molecule has is to know that a double bond is comprised of 1 sigma and 1. The overlap does not occur between the nuclei of the atoms, and this is the key. The molecular geometry of any compound is based on the arrangement of atoms, electron pairs, and bonds.
Not all pi bonds, such as benzene rings and carbon dioxide, are weak bonds in general, you can think of π bonds. A pi bond is a type of covalent bond that exists between atoms where the. Therefore, co2 has 2 pi bonds and 2 sigma bonds.
To start, we must explain. C6h6, c6h12, ch2cl2, ch2 = c = ch2, ch3no2, hconhch3. Sigma and pi bonds are chemical covalent bonds.
This is because among covalent bonds, the pi bond is not a strong bond. My response to, what's a sigma and a pi bond? Sigma and pi bonds are formed by the overlap of atomic orbitals.
It has 2 sigma bonds and 2 pi bonds. Of course the oxygen atoms are equivalent by resonance, and ∠o −o −o = 116.8∘, reduced from the ideal 120∘ angle because. How many sigma and pi bonds in h2so4?
Basics of sigma and pi bonds. One sigma bond and one pi. Sigma and pi bonds are used to describe some features of covalent bonds and molecules with three or two atoms.these bonds are.
Well i think that there are 5 bonds. 3 sigma bonds and 2 pi bonds. Sigma bonds are the product of head on approach of one atomic orbital to other.
Click here👆to get an answer to your question ️ indicate the σ and pi bonds in the following molecules. Since the last 4 electrons are in the pi molecular orbitals, both. How can i identify them in a chemical structure?
C2h2 (ethyne or acetylene) hcch, has 2 sigma bonds between the carbon atoms and the terminal hydrogen atoms, these are single bonds. How many sigma and pi bonds are in a c2 molecule? We can see the 2 × o σ bonds, and the π bond.
One sigma bond and one pi bond combine to form a double bond. Sigma bond is very strong bond due to high extent of overlapping between bonding orbitals.