(the atomic mass of 222 rn 222 rn is 222.01757 u and that of. The equation for the alpha decay of 239pu is:
The isotope 218/84po can decay by either or emission.

The alpha decay of 218 84po. The alpha particle, structurally equivalent to the nucleus of a helium atom and. Let's write out the reaction. Um, from both well, from first the alfa decay process and next, the beta negative decay process for an alfa decay.
In alpha decay, an unstable nucleus will emit an alpha particle, which is a helium nucleus composed of two protons and neutrons. The parent adam here is ah,. The mass of 218/84po, 214/82pb, 4/2he and 218/85at is 218.008973 u, 213.999798 u,.
What is the energy released in this alpha decay reaction 222 86rn→218 84po+4 2he 86 222rn→84 218po+2 4he? What is the nuclear decay equation for an example of alpha decay? This decreases the total mass number by 4 and the atomic number by 2.
Answer 8.6 × 10 − 7 upgrade to view answer discussion. Problem 56 medium difficulty (ii) use fig. Alpha decay is the loss of two protons and two neutrons from a nucleus.
What is the alpha decay of 218 84 po? In an experiment starting with 1gm atom of pure po 218 ,how much time would be. What is the energy release in each case?
Alpha decay is the nuclear decay process whereby the parent nucleus emits an alpha particle. Using the table of masses the po 218 has a mass of 218.008973 and the pb.
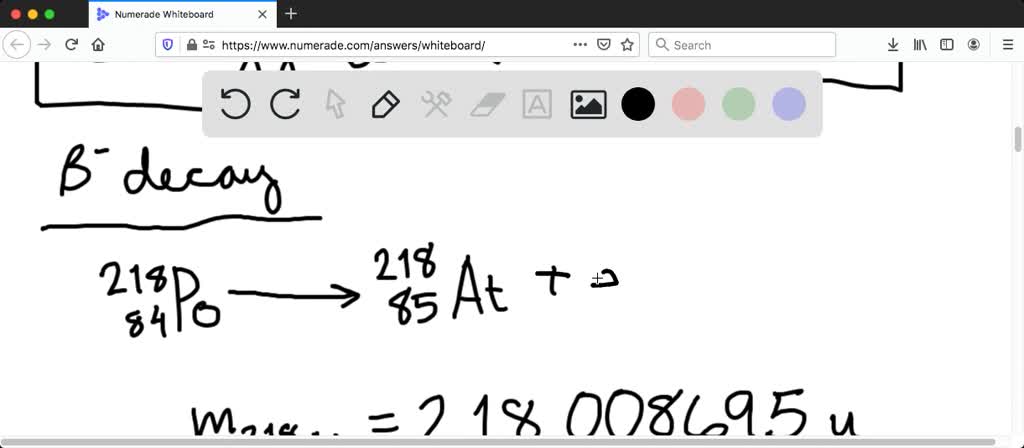
Solved:(Ii) The Isotope ^218_84Po Can Decay By Either \Alpha Or \Beta^- Emission. What Is The Energy Release In Each Case? The Mass Of ^218_84Po Is 218.008973 U.

