Sf4 lewis structure lewis structure is a pictorial representation of the bonds and valence electrons in the molecule. Tell me about the best lewis structure.
What Is The Molecular Geometry Of Brf4- ? A) Square Planar B) Square Pyramidal C) Seesaw D) Tetrahedral | Socratic
Atoms tend to form bonds in order to complete their octet and become stable.

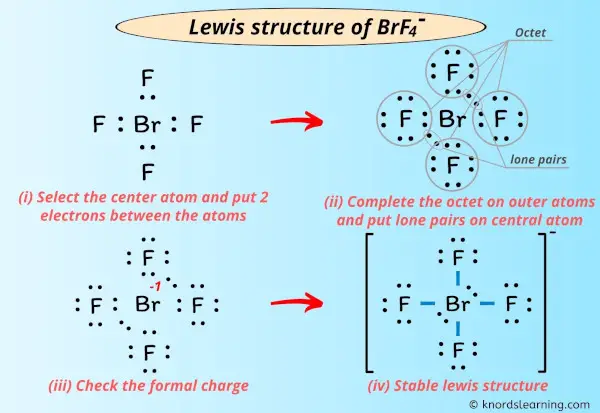
Lewis structure of brf4+. To start, br atom has 7 valence electrons. Vsepr theory predicts this structure as there are 6 electron pairs (4 bonding and 2 lone pairs). The other 4 come from the f atoms.
In the correct lewis structure for brf4+, (br in middle), how many lone pairs (unshared pairs) are on the central atom? This problem has been solved! So to understand the lewis dot structure of brf3, let’s first know some basic details needed to make this structure.
The electron pairs are octahedrallly disposed around. So we have 7 electrons from br, 4 from the f atoms, total 11. Fill in the blank aaf78d029036f9f_1.
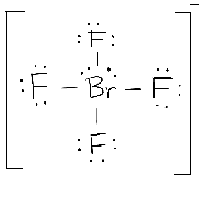
The bonds formed between two atoms are depicted using lines, whereas the valence electrons not forming any bonds are shown by dots. Start from the lewis structure of the tetrafluoroborate ion, brf − 4. Lewis structure is the diagrammatic form given to the skeleton of any molecular composition or ion formed with the help of the constituent elements, the valence electron concept, and the bond formation.
When you do the vsepr you get 6pairs of electron pairs and two lone pairs which means that when you look at the geometrical shape (by looking at how many pairs of electrons you. Sibr4 is a colorless liquid with a very suffocating odor. Lewis structure also relies on this rule, as the atoms in a molecule share their valence electrons and form bonds in order to complete their octet.
Let’s draw and understand this lewis dot structure step by step. The molecule will have a total of 36 valence electrons −7 from bromine, 7 from each of the four fluorine atoms, and one extra electron to given the ion the −1 charge. The bromine atom (br) is at the center and it is surrounded by 4 fluorine atoms (f).
The bromine atom will be bonded to each of the four fluorine atoms via single bonds for a total of 8 of the 36 valence electrons. Once we know how many valence electrons there are in brf4+ we can distribute them around the central atom with the goal of filling the outer shells of each atom. It is synthesized from silicon and vapors of bromine at high temperatures (t>600°c).
4 of those electrons come from br and have already been counted. The answer is a) square planar. Follow some steps for drawing the lewis dot structure of brf5 1.
Experts are tested by chegg as specialists in their subject area. Brf5 lewis structure comprises five fluorine (f) atoms and one bromine (br) atom. Tell me about the atomic charges, dipole moment, bond lengths, angles, bond orders, molecular orbital energies, or total energy.
Hence, sf4 has a trigonal bipyramidal molecular geometry. The halogen group families, which are the 17th group in the periodic table, are both made up of bromine and fluorine atoms. The bromine (br) atom is kept at the central position and the fluorine (f) atoms are in the surrounding position.
We review their content and use your feedback to. Brf5 lewis dot structure by counting valence electrons on the bromine atom to calculate the valence electron of each atom in brf5, look for its periodic group from the periodic table. Lewis structure of the tetrafluoroborate ion, brf 4−.
In the lewis structure of brf4+.

File:solid State Structure Of The (Brf4)+ Ion.png - Wikipedia