It is a polar solvent and is also known as wood alcohol because it was once produced by the distillation of wood. Be notified when an answer is posted.
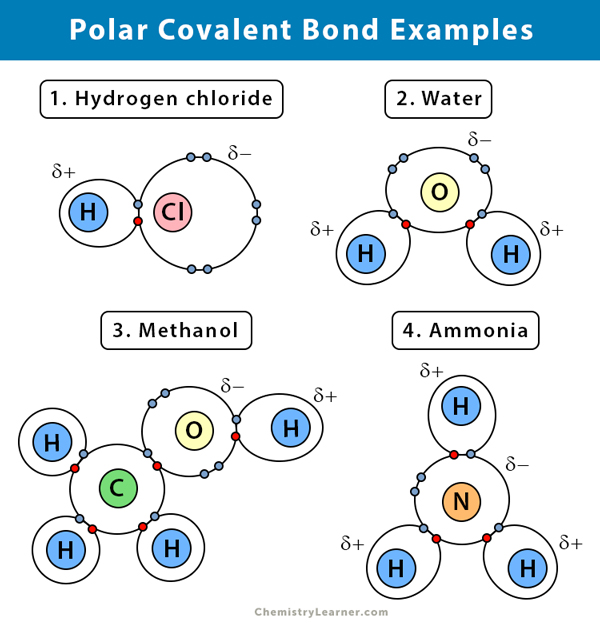
Polar Covalent Bond: Definition And Examples
The molecular weight of methanol is 32.04 g/mol.

Do covalent bonds dissolve in methanol. The boiling point of methanol ( ch3oh) is 64.7 °c. This helps us sort answers on the page. That in hexane, and here the difference in their polarity provides a clear and obvious answer.
Polar lipids are sparingly soluble in hydrocarbon solvents, but dissolve readily in more polar solvents such as methanol, ethanol or chloroform. Do covalent bonds dissolve in methanol? Thanks to entropy, everything dissolves in everything, if only in minuscule amounts.
The bond is formed through the overlap of the oxygen atom having lone pair of an electron with the oxygen atom of an adjoining molecule. The smell of this compound is on the sweeter side as compared to ethanol. O and h in the oh shows covalent.
Electron bonded with 1 covalent bond. So carbon is attached to a few h and 1 oh with four covalent bonds. What do lipids dissolve in?
Not all ionic compounds dissolve in water as water can't solvate some ionic compounds hence hard water. Yes it is because of polarity.as polar molecules dissolve polar molecules (eg water a polar molecule dissolving ethanol also a polar molecule).and vice versa (eg tetrachloro methyl a non polar molecule dissolves in benzene also a non polar molecule)this is just a brief explanation but some how u asked and answer your question. Your response is private was this worth your time?
It results from the attractive force between a hydrogen atom covalently bonded to a very electronegative atom such as a n, o, or f atom and another very electronegative atom. You just need to compare solubilities of k i in methanol and hexane, which basically boils down to the comparison of solvation enthalpy in methanol vs. Not by a long shot.
Nearly everything can be a liquid under the correct conditions. Covalent bonds don't dissolve in water, some covalent compounds because of the polarity of the compound can, such as methanol and ethanol.
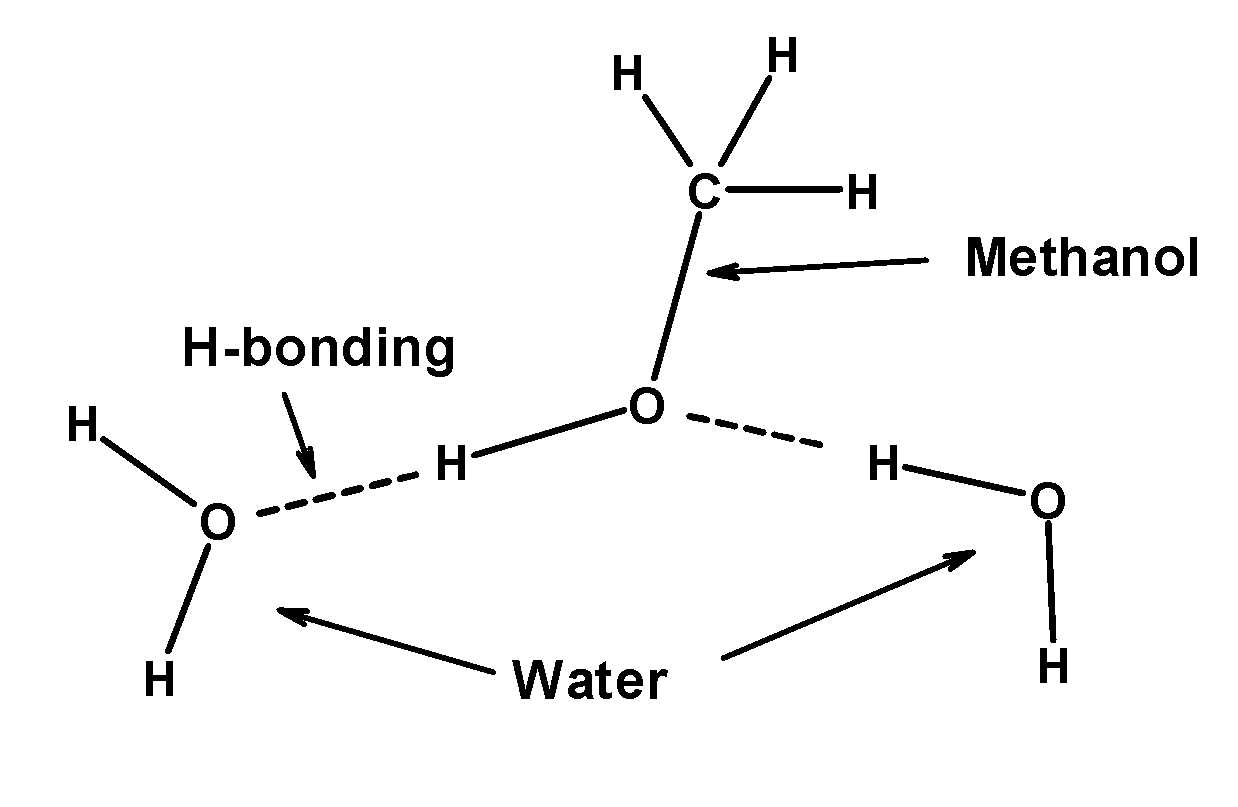
Though Covalent In Nature Methanol Is Soluble In Water Class 11 Chemistry Cbse
What Happens When We Put Covalent Compounds (Solid And Liquid) In Water? Do They Dissolve Just Like Ionic Compounds, By Dissociation? - Quora